34 Spectral Lines and Photons (Graded Reading)
Janice Hester
Emission and Absorption Lines
Most of the light from a star is thermal emission; the dense plasma in the star's photosphere radiates light because of the thermal motions of the electrons and ions making up the plasma. If you look at the spectrum of a star, however, you'll also see dips and spikes at certain wavelengths.
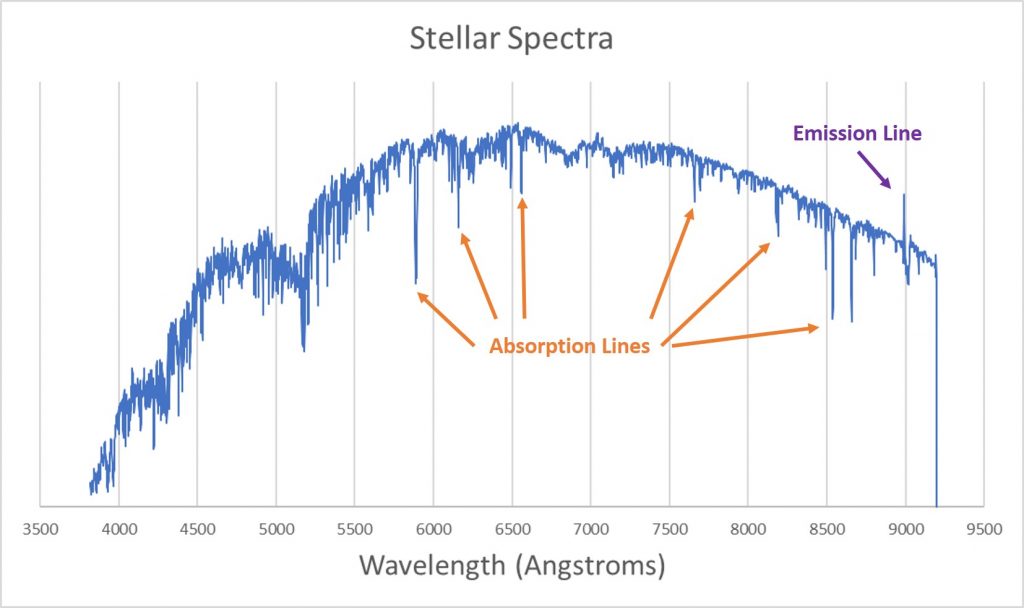
The dips are called absorption lines; they’re wavelengths at which light is missing from the underlying thermal spectrum. The spike (there is only one in this spectrum) is called an emission line; at this wavelength there is extra light that isn’t part of the thermal spectrum.
When a fine grating is used to spread out the Sun’s spectrum, many absorption features are visible (dark bands in the image below).
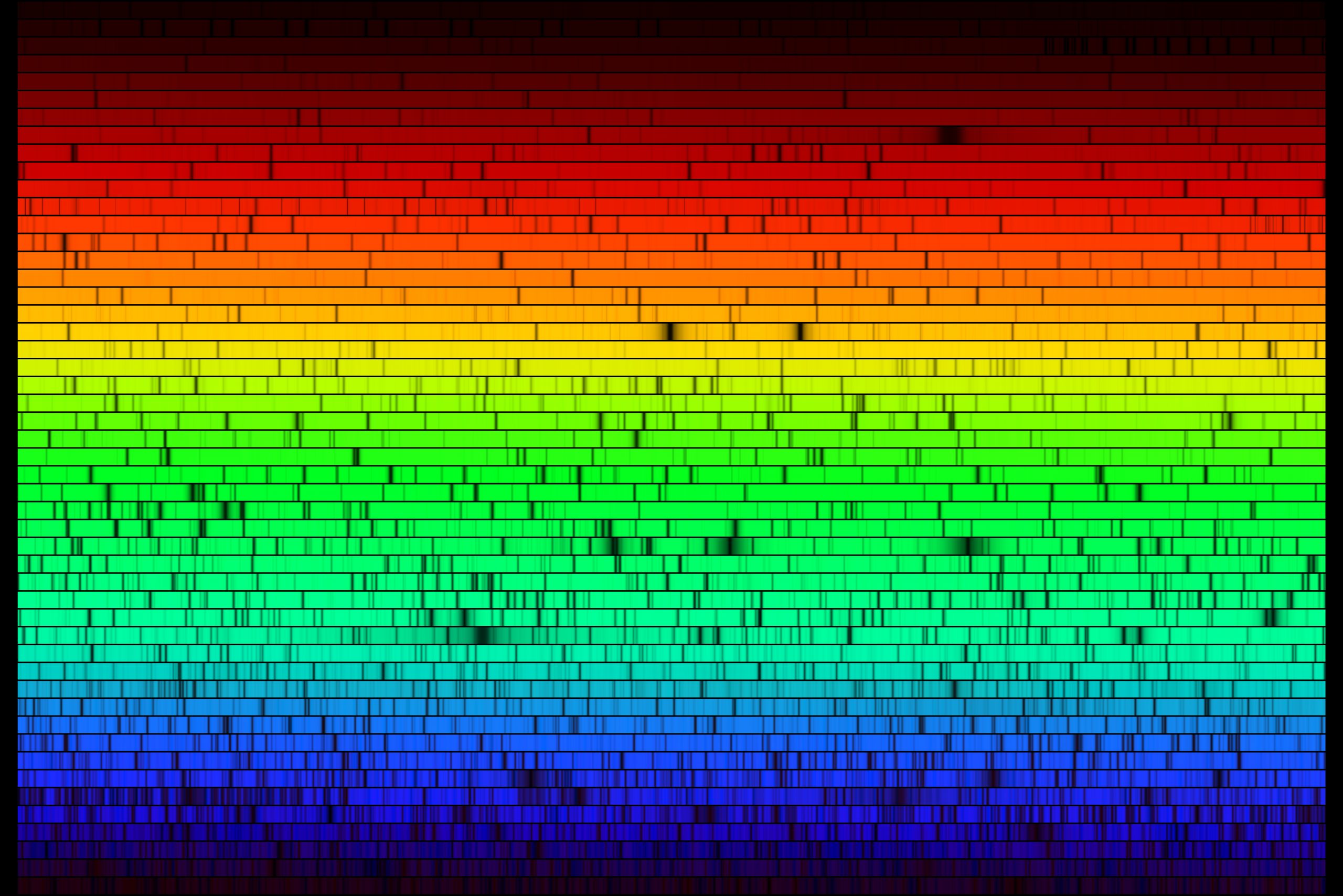
These lines aren’t unique to stellar spectra. As sunlight passes through the Earth's atmosphere, the atmosphere absorbs light of certain wavelengths. This is why space telescopes are needed to observe in most wavelengths outside of visible light. (Radio waves also pass easily through the Earth's atmosphere, which is why radio broadcasts easily travel from the broadcast antenna to a radio's receiving antenna.)
Photons
Emission and absorption lines -- both the wavelengths that they occur at and their shapes (depth/height and width) -- carry a lot of information about the atmospheres responsible for creating them. To understand how they are formed, which is the first step towards learning from them, we need to understand both the nature of light and the nature of atoms and molecules.
The question is why only some some wavelengths of light are absorbed, while other wavelengths of light pass easily through the atmosphere of a particular star or planet. The first part of the answer to this question is photons. Photons are quanta, or packets, of light energy. (Individual photons typically have energies somewhere between about [latex]10^{-30}[/latex] joules up to about [latex]10^{-12}[/latex] joules.)
Whenever light is created or destroyed, it is created and destroyed as photons. Once a photon is created, it can be destroyed (e.g., absorbed), but it cannot be broken apart.
Photons and the Electromagnetic Spectrum
The photons of different colors (wavelengths or frequencies) of light have different energies. The higher the frequency of the light (the shorter the wavelength), the higher the energy of the photons. Blue light is made up of higher-energy photons than red light, and UV photons have higher energies than blue photons.
Photons are the reason that visible light is harmless, UV light can give you a sunburn, too many X-rays can cause cancer, and too many gamma-rays can be fatal. When a photon is destroyed, it deposits all of its energy in one place. The energy carried by an X-ray photon or a gamma-ray photon can damage your tissue; a single photon of visible light can’t.
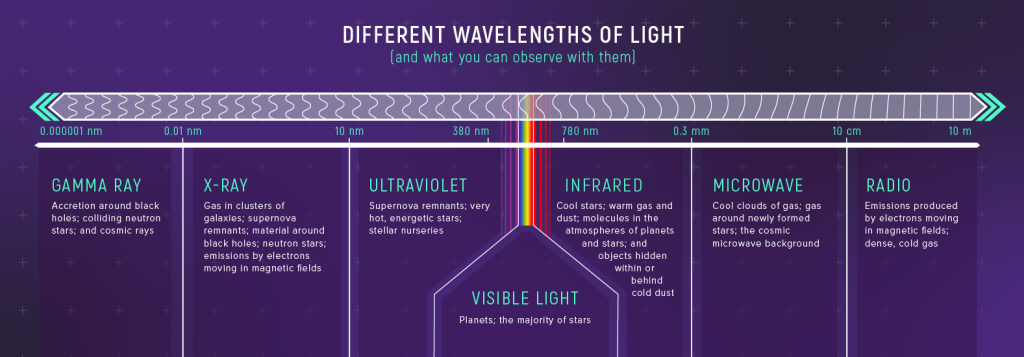
The relationship between energy and wavelength is also why astronomers need telescopes across the full electromagnetic spectrum. High energy objects (accretion disks around black holes, very hot gas, neutron stars, etc.) emit short wavelength light (X-ray and gamma-ray) made up of high-energy photons. The coolest stars and the warm gas surrounding star forming regions emit in the infrared, and the coldest gas emits at even longer wavelengths. (The connection between photon energy and wavelength is why hotter objects emit at shorter wavelengths.)
Planck's Constant
The formula relating the frequency of a light wave to the energy of its photons is
[latex]E=hf[/latex]
In this formula, [latex]f[/latex] is the frequency of a light wave and [latex]E[/latex] is the energy of a single photon of light. The constant [latex]h[/latex] is called Planck's constant. The energy of a single photon is directly proportional to the frequency of the light wave.
Recall that for light traveling through a vacuum [latex]c=\lambda f[/latex] ([latex]c[/latex] is the speed of light through a vacuum and [latex]\lambda[/latex] is the wavelength). The formula relating the energy of a single photon and the wavelength of the light wave is
[latex]E=\frac{hc}{\lambda}[/latex]
The energy of a single photon is inversely proportional to the wavelength of the light wave.
The longest wavelength UV light has a wavelength of about 350 nm; red light has a wavelength of about 700 nm. Since the wavelength of this UV light is about half the wavelength of red light, the UV photons each carry about twice as much energy.
When a star's atmosphere absorbs light of a particular wavelength, it is absorbing photons of a particular energy. To understand why only photons of particular energies are absorbed (or created in the case of emission lines), we need to need to switch to discussing atoms and molecules.
