2 Intermolecular Forces
Chapter 2 Learning Objectives
- Be able to state and define the four types of intermolecular forces.
- Be able to arrange the intermolecular forces based on their strength.
- Be able to predict physical properties such a smelting point, boiling point and solubility of various types of organic compounds based on the intermolecular forces present.
2.1 Introduction
The physical properties of a compound are determined by the forces between the individual molecules known as intermolecular forces. Understanding how intermolecular forces operate allows us to compare compounds to each other in a relative way. By applying predictable trends trends, we can, for example, identify which compound will boil at a higher temperature. A practical example of this is the process of fractional distillation. Crude oil is an important natural resource. Different parts (fractions) of crude oil are used to make fuels, plastics, medicines, fabrics, and dyes. Crude oil is separated by fractional distillation. In this process, the crude oil is heated up until different fractions of it boils. The fractions are cooled and collected. This process is effective because each fraction has a different boiling point. Smaller molecules with weaker intermolecular forces are easier to separate and boil at lower temperatures. Larger molecules with very strong intermolecular forces require significantly more energy to separate and have higher boiling points. This same concept also extends to other properties such as melting point, volatility, viscosity, surface tension and solubility in polar and nonpolar solvents.
To understand intermolecular forces between molecules, one must have a good knowledge of chemical bonding. If you need to review important aspects of chemical bonding, visit this site. Intermolecular forces are forces between molecules. It is important to note that they are different, but dependent upon intramolecular forces which are forces within molecules.
2.2 Types of Intermolecular Forces
We will examine four types of intermolecular forces:
- Ion-dipole/ionic: The ion-dipole force is not typically categorized as an intermolecular force, as it is an important non-covalent force that is responsible for the interaction between ions and other polar substances. A simple example is the dissolving of an ionic solid, or salt, in water. When table salt (NaCl) is dissolved in water, the interactions between the ions and water molecules are strong enough to overcome the ionic bond that holds the ions in the crystal lattice. As a result, the cations and anions are separated completely, and each ion is surrounded by a cluster of water molecules. This is called a solvation process. The solvation occurs through the strong ion-dipole force. Many salts, or ionic compounds, are soluble in water because of such interactions.
- Hydrogen bonding: First of all, do not let the name mislead you! Although it is called a “bond”, a hydrogen bond is not a covalent bond, it is a type of intermolecular force. The hydrogen bond is the force between an H atom that is bonded to O, N or F (atoms with high EN) and the neighboring electronegative atom.
- Dipole-dipole: For polar molecules, molecules are attracted to each other because of a permanent dipole, this type of attractive force is called a dipole-dipole force. As shown below in the electrostatic potential map of acetone, one end of acetone has a partial negative charge (red), and the other end has a partial positive charge (blue). The dipole-dipole force is an attraction force between the positive end of one molecule and the negative end of the neighboring molecule.
- London dispersion forces: Dispersion forces (also called London forces) result from the instantaneous dipole and induced dipole of the molecules. For nonpolar molecules, the constant shifting and distortion of electron density leads to a weak, short-lived dipole at a given moment, which is called an instantaneous dipole. Such temporary dipoles also induce the electrons in a neighbouring molecule to be distorted, and this molecule develops a corresponding transient dipole of its own, which is the induced dipole. At the end, all nonpolar molecules are attracted together via the two types of temporary dipoles. The dispersion force is weak in nature and is the weakest intermolecular force. However, since it applies to all types of molecules (it is the only intermolecular force for nonpolar molecules), dispersion forces are also the most fundamental intermolecular force.
The magnitude of dispersion forces depends on two factors:
- The relative polarizability of electrons – The simple understanding of polarizability is how easily the electrons get distorted. For larger atoms, there are more electrons in a larger space; therefore the electrons are more loosely held and more easily polarized, so the dispersion force is stronger. Generally, the larger the molar mass of the molecule, the stronger the dispersion force.
- The relative surface area of the molecule – Molecules with longer, flatter or cylindrical shapes have a greater surface area compared to the bulky, branched molecules, and therefore have a stronger dispersion force. Taking the two constitutional isomers of C4H10, butane and isobutane as an example, the dispersion force of butane is stronger than that of isobutane.
These intermolecular forces are listed from strongest (1) to weakest (4). Visit this site for a detailed explanation of each of these forces, worked examples and additional practice problems.
Credits: Lumen Learning provides a simple, supported path for faculty members to adopt and teach effectively with open educational resources (OER). Unless otherwise noted, content on this site is licensed under the Creative Commons Attribution 4.0 License.
A notable instance of hydrogen bond can be found in the DNA backbone. See how the base pairs hydrogen bond and hold the two strands together (Figure 2.1).
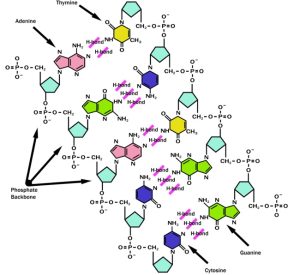
Figure 2.1: Hydrogen Bonding in DNA
Credits: This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
2.3 Effect of Intermolecular Forces on Boiling Points and Melting Points
We will now examine how intermolecular forces affect boiling point. This video gives an overview of intermolecular forces on boiling points. This site examines how intermolecular forces can be used to predict melting and boiling point trends.
The b.p. trend of different substances directly correlates with the total intermolecular forces. Generally speaking, the stronger the overall intermolecular force applied to a certain substance, the higher the b.p. of the substance. The b.p. is the temperature at which the liquid phase of the substance vaporizes to become a gas. To vaporize a liquid, the intermolecular forces that hold the molecules together must be overcome. The stronger the forces, the more energy that is needed to overcome the forces, and a higher temperature is required, thus leading to a higher b.p.
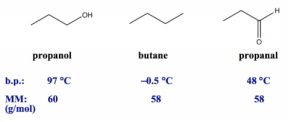
Figure 2.2: Boiling Points of Compounds with Similar Masses
All three compounds in Figure 2.2 have similar molar masses, so the dispersion forces are at a similar level. However, the three compounds have different molecular polarities. Butane is a non-polar substance that only has dispersion forces, propanal is a polar molecule with both dispersion forces and dipole-dipole forces, and propanol is a polar molecule with an OH bond, so all three types of forces apply. Therefore, the overall amount of intermolecular force is strongest for propanol and weakest for butane, which is in the same order as their boiling points.
Key Takeaways: Influence of IMF on Melting and Boiling Points
- Stronger the IMF, higher are the boiling & melting points of organic molecules.
- Greater the number of IMF, higher are the boiling & melting points.
- Symmetrical structures have a higher melting point than less symmetrical structures.
2.4 Effect of Intermolecular Forces on Solubility
Intermolecular forces can also be used to predict solubility. The stronger the intermolecular forces between solute molecules and solvent molecules, the greater the solubility of the solute in the solvent. Do you remember the rule, “like dissolves like”. A general rule for solubility is summarized by the expression “like dissolves like”. This means that one substance can dissolve in another with a similar polarity and, as a result, with similar intermolecular forces.
More specifically:
- Nonpolar substances are usually soluble in nonpolar solvents.
- Polar and ionic substances are usually soluble in polar solvents.
- Polar and nonpolar substances are insoluble to each other.
Water, methanol and ethanol are examples of very polar solvents that can form hydrogen bonds. Ether, ketone, halide and esters are polar solvents as well, but they are not as polar as water or methanol. Non-polar solvents include hydrocarbons like hexane, benzene, and toluene. For large organic molecules, however, it may not be so easy to simply call them polar or non-polar because part of a compound may be polar while another part may be nonpolar. Such polar or nonpolar parts of a compound are often described as hydrophilic or hydrophobic.
- Hydrophilic (hydro, water; philic: loving or seeking) means it likes water, or is soluble in water.
- Hydrophobic (hydro, water; phobic: fearing or avoiding) meaning it does not like water, or is insoluble in water.
The hydrocarbon part of an organic compound is hydrophobic because it is nonpolar and therefore does not dissolve in polar water. The functional group of OH, COOH, NH2, etc. is polar and is therefore hydrophilic. With both hydrophobic and hydrophilic parts present in an organic compound, the overall polarity depends on whichever part is the major one. If the carbon chain is short (1~3 carbons), the hydrophilic effect of the polar group is the major one, so the whole compound is soluble in water; with carbon chains of 4~5 carbons, the hydrophobic effect begins to overcome the hydrophilic effect, and water solubility is lost.
Key Takeaways: Influence of IMF on Solubility
- Ionic compounds and polar molecules are soluble in polar solvents.
- Nonpolar molecules are soluble in nonpolar solvents.
- Stronger the IMF, higher is the solubility of organic molecules.
- Greater the number of IMF, higher is the solubility of polar molecules in polar solvents.
Additional Resources:
Fundamentals of Intermolecular Forces
Full Lecture on Intermolecular Forces
Additional Intermolecular Forces Review
Van der Waals Forces Explained
Intermolecular Forces and Solubility
Boiling Points of Organic Compounds
Exercise: Test your knowledge in IMF
Exercises: Watch this interactive video to test your knowledge in IMF.
Key Takeaways: IMF
- Intermolecular forces occur between molecules. They play a vital role in the physical properties of the compound such as boiling point, melting point and solubility.
- There are three main types of IMF: dispersion forces, dipole forces and hydrogen bonding.
- Strength of IMF: Dispersion forces < Dipole forces < Hydrogen bonding
- Greater the number of intermolecular forces, greater is the melting and boiling point.
- Compounds that are capable of hydrogen bonding are soluble in water.

