MIXED UNKNOWN
LEARNING OBJECTIVES
Demonstrate competency of microbiology laboratory techniques by separating and identifying two organisms in a mixed broth
Design two dichotomous keys to differentiate the possible Gram-negative unknowns and the possible Gram-positive unknowns
MCCCD OFFICIAL COURSE COMPETENCIES
Describe the modes of bacterial and viral reproduction and proliferation
Utilize aseptic technique for safe handling of microorganisms
Apply various laboratory techniques to identify types of microorganisms
Identify structural characteristics of the major groups of microorganisms
Compare and contrast prokaryotic cell and eukaryotic cell
Compare and contrast the physiology and biochemistry of the various groups of microorganisms
MATERIALS
Unknown mixed broth containing Gram-positive cocci and Gram-negative bacilli used for 2 lab sections
1 MacConkey Agar
1 CNA Agar
Inoculating loop
Sterile swab
BIOCHEMICAL TESTS ALBUM LINK
Possible Gram-negative unknowns: Possible Gram-positive unknowns
Pseudomonas aeruginosa Staphylococcus aureus
Alcaligenes faecalis Staphylococcus epidermidis
Escherichia coli Micrococcus luteus
Klebsiella aerogenes Micrococcus roseus
Proteus mirabilis Group C Streptococcus
Klebsiella pneumoniae Streptococcus pyogenes
Salmonella enterica Streptococcus pneumoniae
Shigella flexneri Streptococcus sanguinis
Enterococcus faecalis
There are two different types of bacteria in the broth of the mixed unknown. One type is Gram-positive cocci and the other is Gram-negative bacilli. First, you must isolate and separate the two organisms from each other by using selective media.
The MacConkey Agar is selective for Gram-negative organisms. The crystal violet dye and the bile salts in the media inhibit the growth of Gram-positive organisms. MacConkey agar is also a differential media because it contains lactose. Gram-negative bacilli that ferment lactose will appear hot pink and those that do not ferment lactose will appear colorless.
The Colistin-Naladixic Agar is selective for Gram-positive organisms. CNA contains the antibiotics colistin and naladixic acid which suppress the growth of Gram-negative organisms. CNA is differential because it contains red blood cells. You can determine the hemolytic properties of the Gram-positive cocci.
Once the bacteria have been isolated from each other, the next step is to identify both bacteria. By using biochemical test charts to differentiate the Gram-positive cocci and the Gram-negative bacilli, you will design two dichotomous keys (flow charts) to guide you in determining which tests will help you identify your unknowns. The result of the first test will determine the next test to perform.
You must complete the tests in the order of the dichotomous key until you reach an identification of your unknowns. Record the biochemical tests you perform in the order of your dichotomous key. Record the identification of the unknown using correct binomial nomenclature.
PRE-ASSESSMENT
PROCEDURE
Day 1: streak plates of selective/differential media
streak plate
1. Label the MacConkey and CNA plates with your name (not initials), date, unknown number and the name of the media. Record your unknown number on the worksheet.
2. Divide the MacConkey and CNA plates into 3 sections by drawing a “T” on the bottom of the plate where the media is.
 3. Resuspend the mixed unknown broth culture by gently shaking or tapping the tube side to side until the bacteria swirl up from the bottom of the tube.
3. Resuspend the mixed unknown broth culture by gently shaking or tapping the tube side to side until the bacteria swirl up from the bottom of the tube.
MacConkey Agar Streak Plate
1. Peel the paper cover from the wooden end of a sterile swab. Handle ONLY the wooden end of the swab to maintain the sterility of the cotton end.
2. Holding the tube in your non-dominant hand, remove the cap of the bacterial culture with your little finger on your dominant hand. Dip the cotton end of the swab into the mixed unknown broth and then slightly press the swab against the inside of the tube to expel any excess broth. Recap the bacterial culture tube and place it in a test tube rack.
3. Using your non-dominant hand, open the lid of the plate just enough to fit the swab in.
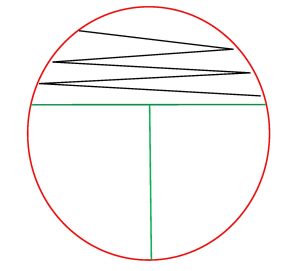
4. Inoculate the first section of the plate by touching the swab down on the surface of the agar and drawing a zig-zag streak to fill that first area of the plate. Replace the Petri plate lid. Dispose of the swab in the biohazard trash.
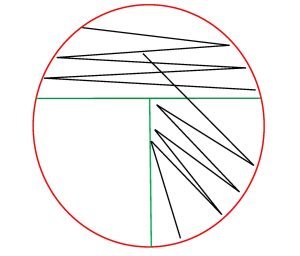
5. Use an inoculating loop to complete the rest of the procedure.
6. Sterilize your inoculating loop in the Bacticinerator and allow it to cool.
7. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the first section and gently drag the loop one time into the second section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the second section. Replace the Petri plate lid.
 8. Sterilize the loop in the Bacticinerator and allow it to cool.
8. Sterilize the loop in the Bacticinerator and allow it to cool.
9. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the second section and gently drag the loop one time into the third section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the third section. Replace the Petri Plate lid.
10. Sterilize the loop in the Bacticinerator and allow it to cool.
11. Incubate your MacConkey agar plate lid side down in the class plate tray until the next lab period.
CNA Agar Streak Plate
1. Peel the paper cover from the wooden end of a sterile swab. Handle ONLY the wooden end of the swab to maintain the sterility of the cotton end.
2. Holding the tube in your non-dominant hand, remove the cap of the bacterial culture with your little finger on your dominant hand. Dip the cotton end of the swab into the mixed unknown broth and then slightly press the swab against the inside of the tube to expel any excess broth. Recap the bacterial culture tube and place it in a test tube rack.
3. Using your non-dominant hand, open the lid of the plate just enough to fit the swab in.

4. Inoculate the first section of the plate by touching the swab down on the surface of the agar and drawing a zig-zag streak to fill that first area of the plate. Replace the Petri plate lid. Dispose of the swab in the biohazard trash.

5. Use an inoculating loop to complete the rest of the procedure.
6. Sterilize your inoculating loop in the Bacticinerator and allow it to cool.
7. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the first section and gently drag the loop one time into the second section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the second section. Replace the Petri plate lid.
 8. Sterilize the loop in the Bacticinerator and allow it to cool.
8. Sterilize the loop in the Bacticinerator and allow it to cool.
9. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the second section and gently drag the loop one time into the third section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the third section. Replace the Petri Plate lid.

10. Sterilize the loop in the Bacticinerator and allow it to cool.
11. Stab the sterilized loop 2–3 times into the agar in the first section of the streak plate. Stabbing into the media will enhance the appearance of hemolysis.
12. Incubate your CNA plate lid side down in a candle jar since some of the unknown Gram-Positive organisms are microaerophilic.
13. Label the side of your mixed unknown broth with a permanent marker with your name and lab section (day & time). Place the broth in the rack. These tubes will be saved in the refrigerator in case your unknowns do not grow on the CNA or MacConkey agar plates.
Day 2: Make Stock Cultures
gram-negative bacilli unknown
MACCONKEY AGAR AFTER INCUBATION – Observe your MacConkey Agar plate and determine if your organism ferments lactose and record your results on the mixed unknown worksheet. If you do not have growth, reincubate your plates and subculture the original broth again.
Hot pink colonies (look at the color of the colonies not the color of the media) = Positive for lactose fermentation
Clear colonies = Negative for lactose fermentation (some lactose negative colonies may have a slight pink color due to the transmittance of the media color through the colony)

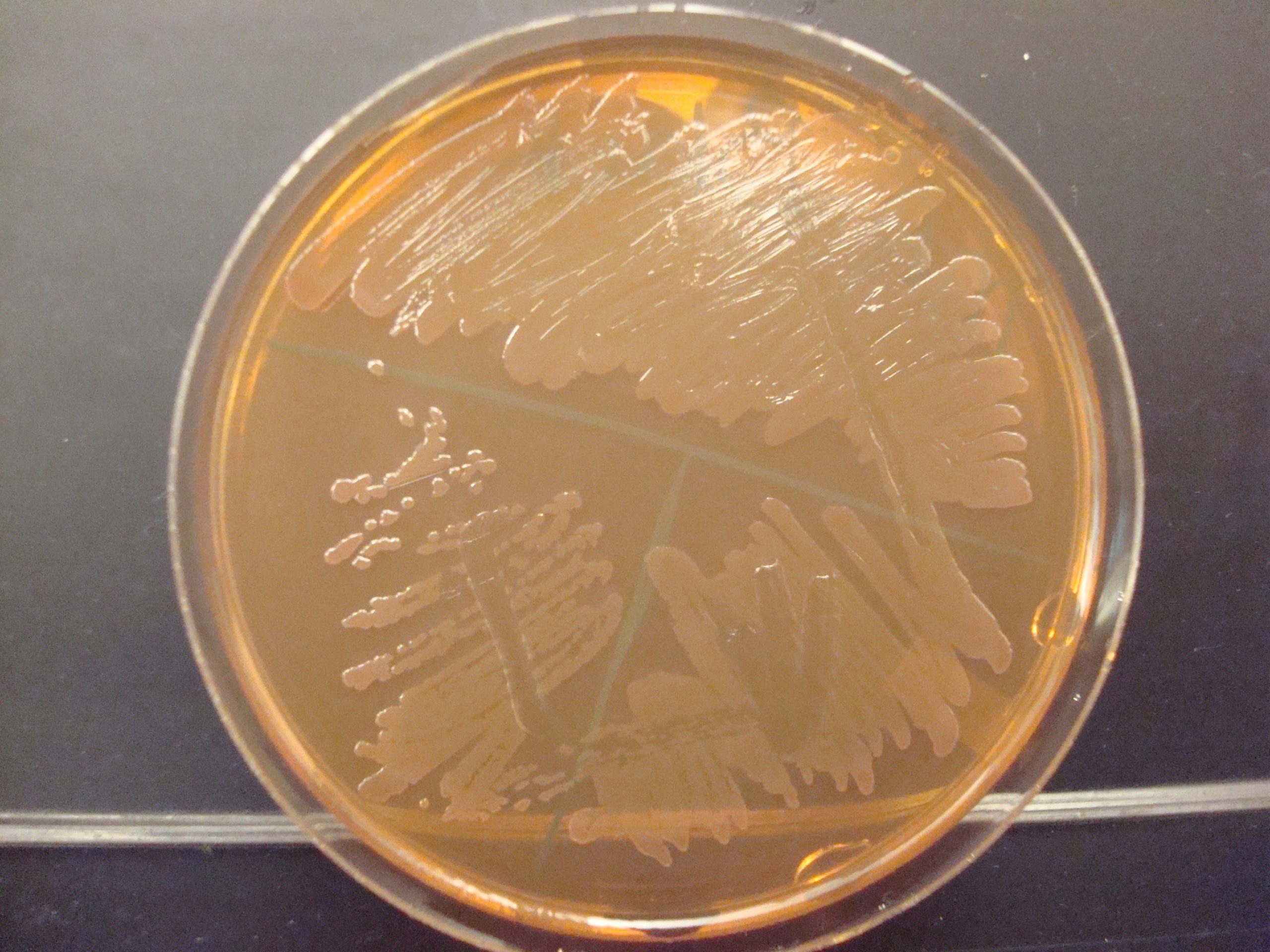
MAKE THE GRAM-NEGATIVE STOCK CULTURE streak plate
1. Label a TSA plate with your name, your unknown number, and the words “Gram Negative Stock”. Divide the plate into 3 sections by drawing a “T” on the plate. THIS WILL BE YOUR GRAM NEGATIVE STOCK CULTURE. You will use this stock culture to inoculate all future biochemical tests performed on this unknown. TSA is a “non-inhibitory” media which allows the organism to produce enzymes and metabolize the ingredients in the media.

2. Sterilize your inoculating loop in the Bacticinerator and allow it to cool thoroughly.
3. Use the inoculating loop take ONE SINGLE ISOLATED colony from the MacConkey Agar.
4. Open the lid of the TSA plate just enough to fit the loop in. Inoculate the first section of the plate by touching the loop down on the surface of the agar and drawing a zig-zag streak to fill that first area of the plate. Replace the Petri plate lid.

5. Sterilize your inoculating loop in the Bacticinerator and allow it to cool.
6. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the first section and gently drag the loop one time into the second section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the second section. Replace the Petri plate lid.

7. Sterilize the loop in the Bacticinerator and allow it to cool.
8. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the second section and gently drag the loop one time into the third section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the third section. Replace the Petri Plate lid.

9. Sterilize the loop in the Bacticinerator and allow it to cool.
10. Incubate the Gram-Negative TSA stock plate lid side down in the class plate tray until the next lab period.
11. Place the MacConkey plate lid side down in the class tray to be saved in the refrigerator in case your Gram-Negative stock plate does not grow.
GRAM-POSITIVE COCCI UNKNOWN
AFTER INCUBATION – Evaluate the type of hemolysis produced by the organisms. Hold the plate so that light is shining through the back of the agar. Determine the extent of damage to the blood in the agar surrounding the bacterial colonies. If condensation is preventing you from seeing the hemolysis, lift the lid of the plate just enough to insert a sterile swab. Use the sterile swab to remove the condensation. Record the hemolysis results on the mixed unknown worksheet.
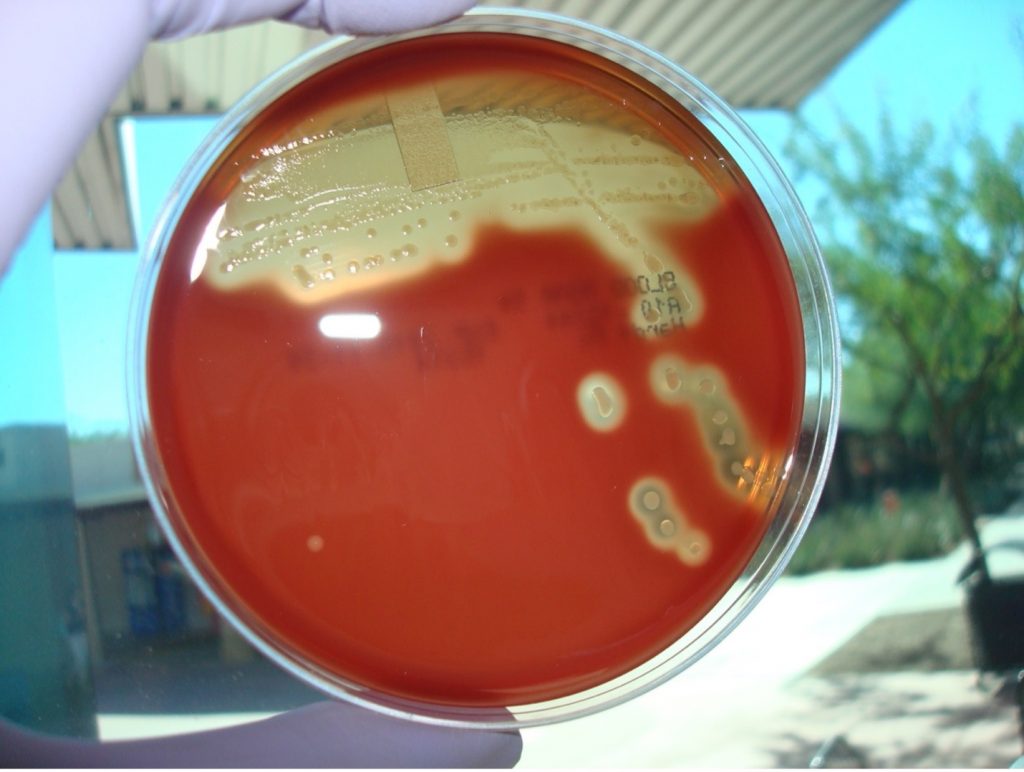
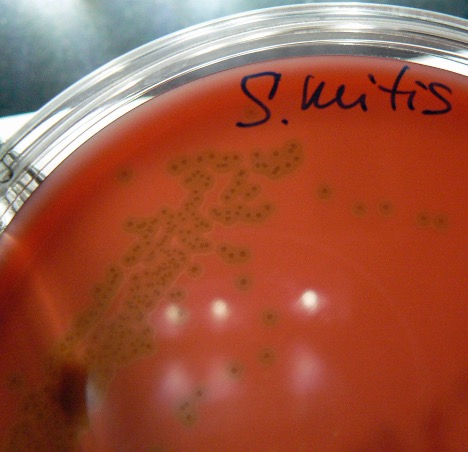
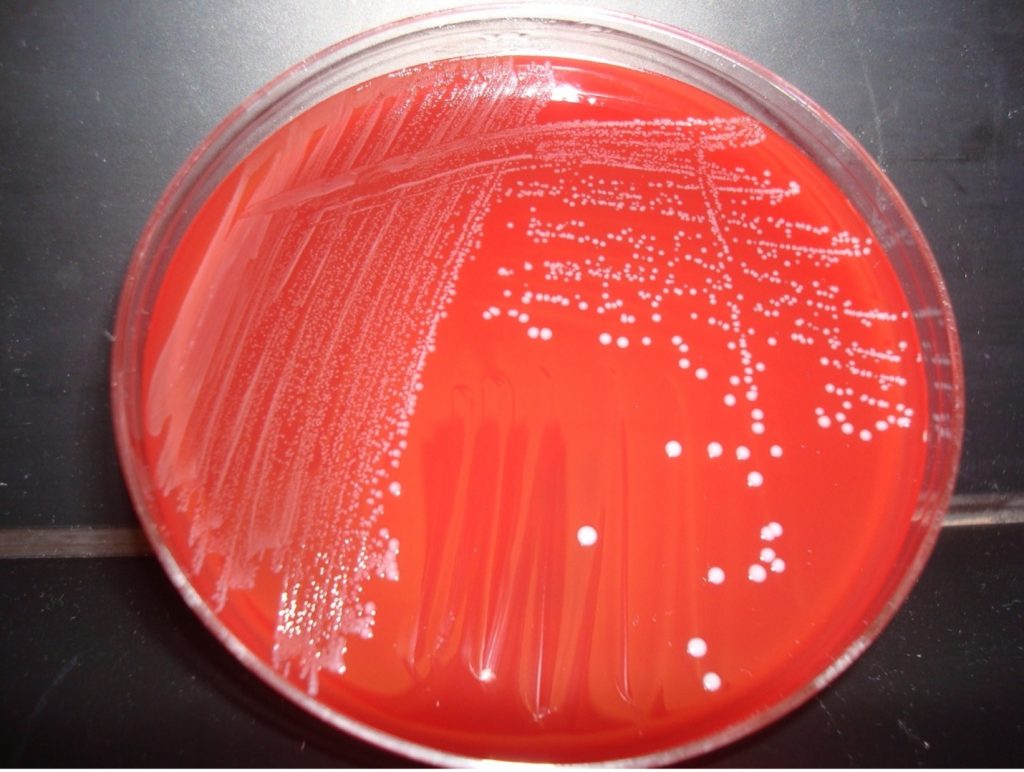
MAKE THE GRAM-positive STOCK CULTURE streak plate
1. Label a TSA plate with your name, your unknown number, and the words “Gram Positive Stock”. Divide the plate into 3 sections by drawing a “T” on the plate. THIS WILL BE YOUR GRAM POSITIVE STOCK CULTURE. You will use this stock culture to inoculate all future biochemical tests performed on this unknown. TSA is a “non-inhibitory” media which allows the organism to produce enzymes and metabolize the ingredients in the media.

2. Sterilize your inoculating loop in the Bacticinerator and allow it to cool thoroughly. Use the inoculating loop take ONE SINGLE ISOLATED colony from the CNA Agar.
3. Open the lid of the TSA plate just enough to fit the loop in. Inoculate the first section of the plate by touching the loop down on the surface of the agar and drawing a zig-zag streak to fill that first area of the plate. Replace the Petri plate lid.

4. Sterilize your inoculating loop in the Bacticinerator and allow it to cool.
5. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the first section and gently drag the loop one time into the second section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the second section. Replace the Petri plate lid.

6. Sterilize the loop in the Bacticinerator and allow it to cool.
7. Open the lid of the plate just enough to fit the loop in. Place the loop in the center of the second section and gently drag the loop one time into the third section. Lightly drag the tip of the loop from side to side in a back-and-forth motion to spread the inoculum to fill the third section. Replace the Petri Plate lid.

8. Sterilize the loop in the Bacticinerator and allow it to cool.
9. Incubate your Gram-Positive Stock Plate lid side down in a candle jar since some of the unknown Gram-Positive organisms are microaerophilic.
10. Place the CNA plate lid side down in a candle jar or the class tray to be saved in the refrigerator in case your Gram-Positive stock culture does not grow.
Day 3: Begin testing your unknown organisms
1. As soon as you have your dichotomous keys graded and your organisms growing on stock cultures, you can begin to perform biochemical tests. YOU MUST PERFORM THE TESTS IN THE ORDER ON YOUR DICHOTOMOUS KEY.
2. Use your stock culture to perform the first test listed on your dichotomous key. Label the side of the test media with a permanent marker with your name, test name, date, and whether the organism is your Gram- Negative or Gram-Positive unknown. Incubate the test until the next lab session.
* You can only inoculate ONE test/unknown/day unless you can determine the results of the biochemical test right away.
3. Work on identifying both unknowns simultaneously. Organize your work so you do not mix up the tests. In your test tube rack, place the stock culture you are using in front of the biochemical test you are going to inoculate. Immediately label the test media with your name, date, media name and whether this is the Gram-Positive or Gram-Negative unknown.
4. Once you have recorded the reactions and results, properly dispose of the test. Do not keep the biochemical tests after you have recorded the results.
5. Once you have identified both unknowns, turn in your mixed unknown worksheet and dichotomous keys. After confirming your results with your instructor, dispose of all your test media, agar plates, and broths.
TESTS RESULTS OF GRAM-POSITIVE COCCI UNKNOWN
TEST RESULTS OF STAPHYLOCOCCUS AND MICROCOCCUS
| TEST | Staphylococcus aureus | Staphylococcus epidermidis | Micrococcus roseus | Micrococcus luteus |
|---|---|---|---|---|
| Catalase | Positive | Positive | Positive | Positive |
| Glucose Fermentation | Positive | Positive | Negative | Negative |
| Coagulase | Positive | Negative | Negative | Negative |
| Salt Tolerance on Mannitol Salt Tolerance | Positive | Positive | Negative | Negative |
| Mannitol Fermentation on Mannitol salt Agar | Positive | Negative | Negative | Negative |
| Pigment Production (colony color) | White | White | Rose-Coral | Yellow |
| Nitrate Reduction (not done in this exercise) | Positive | Positive | Positive | Negative |
TEST RESULTS OF STREPTOCOCCUS AND ENTEROCOCCUS
| TEST | Streptococcus sanguinis | Streptococcus pneumoniae | Streptococcus pyogenes | Group C Streptococcus |
Enterococcus faecalis |
| Catalase | Negative | Negative | Negative | Negative | Negative |
| Bile Esculin | Negative | Negative | Negative | Negative | Positive |
| Hemolysis | Alpha | Alpha | Beta | Beta | *Variable |
| Optochin sensitivity | Resistant (Negative) |
Sensitive (Positive) |
|||
| Latex Agglutination | Positive reaction with Group A antibodies | Positive reaction with Group C antibodies |
NOTE: “Variable” test result means the result is not consistent. Some strains of the species will exhibit gamma hemolysis whereas other strains will produce alpha or beta hemolysis.
TEST RESULTS OF GRAM-NEGATIVE Bacilli UNKNOWN
TESTS RESULTS OF GRAM-NEGATIVE UNKNOWN
| TEST | E.coli | Klebsiella aerogenes |
Klebsiella pneumoniae |
Proteus mirabilis | Salmonella | Shigella | Pseudomonas aeruginosa | Alcaligenes faecalis |
| Oxidase | Negative | Negative | Negative | Negative | Negative | Negative | Positive | Positive |
| Glucose fermentation | Positive | Positive | Positive | Positive | Positive | Positive | Negative | Negative |
| Lactose fermentation | Positive | Positive | Positive | Negative | Negative | Negative | Negative | Negative |
| Sucrose fermentation | Negative | Positive | Positive | Negative | Negative | Negative | Negative | Negative |
| Nitrate reduction | Positive | Positive | Positive | Positive | Positive | Positive | Positive | Negative |
| Urease | Negative | Negative | Positive | Positive | Negative | Negative | Not reliable | Negative |
| Indole | Positive | Negative | Negative | Negative | Negative | Positive | Not reliable | Not reliable |
| MR Test | Positive | Negative | Negative | Positive | Positive | Positive | Negative | Negative |
| VP Test | Negative | Positive | Positive | Negative | Negative | Negative | Negative | Negative |
| Citrate | Negative | Positive | Positive | Positive | Positive | Negative | Positive | Not reliable |
| KIA H2S production | Negative | Negative | Negative | Positive | Positive | Negative | Negative | Negative |
| Motility | Positive | Positive | Negative | Positive | Positive | Negative | Not reliable | Negative |
DISCOVERIES IN MICROBIOLOGY
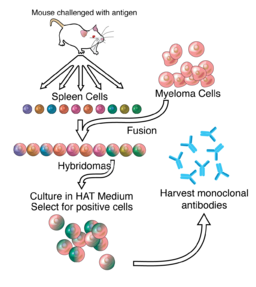 DR. CÉSAR MILSTEIN
DR. CÉSAR MILSTEIN
In 1975, Argentine biochemist Dr. César Milstein was the first to produce monoclonal antibodies. Prior to the development of Dr. Milstein’s technique, scientists had struggled to produce large amounts of single antibody clones because of the difficulty of growing antibody producing B cells in culture. Dr. Milstein overcame this problem by fusing spleen cells derived from mice immunized with a specific antigen to immortalized myeloma cells, a technique that enabled production of antigen-specific antibody in culture indefinitely. His techniques were used to produce diagnostic tests, cancer treatments, disease treatment, vaccines, and blood and tissue typing.

