20 DIFFERENTIATION OF GRAM NEGATIVE BACILLI
LEARNING OBJECTIVES
Distinguish between bacteria belonging in the Family Enterobacteriaceae from non-Enterobacteriaceae
State the purpose and principle of the oxidase test, glucose carbohydrate fermentation tests and KIA
Perform and interpret the oxidase test, nitrate reduction test, glucose carbohydrate fermentation tests and KIA
State the significance of glucose fermentation and oxidase test in identifying Gram-negative bacilli
MCCCD OFFICIAL COURSE COMPETENCIES
Describe the modes of bacterial and viral reproduction and proliferation
Utilize aseptic technique for safe handling of microorganisms
Apply various laboratory techniques to identify types of microorganisms
Identify structural characteristics of the major groups of microorganisms
Compare and contrast prokaryotic cell and eukaryotic cell
Compare and contrast the physiology and biochemistry of the various groups of microorganisms
MATERIALS
Stock Cultures: per group of four students
Escherichia coli on TSA slant
Alcaligenes faecalis on TSA slant
Proteus mirabilis on TSA slant
Pseudomonas aeruginosa on TSA slant
Media: per person
MacConkey agar plate
KIA tube
Nitrate broth
Oxidase strip in open Petri dish
Equipment:
Inoculating loop
Inoculating wire
Sterile swabs
Water dropper bottle
BIOCHEMICAL TESTS ALBUM LINK
Gram negative bacilli comprise a vast array of bacteria, yet most clinically significant Gram-negative bacilli can be divided into two groups.
One major group of Gram-negative bacilli is the Family Enterobacteriaceae which contain many genera of organisms such as E. coli, Klebsiella, Enterobacter and Proteus. These Gram-negative, facultative anaerobes are frequently called “enterics” since they are normally found in the intestinal tract of humans and other animals.
The Gram-negative bacilli that are not in the Enterobacteriaceae family are composed of several families but are collectively called Non-Enterobacteriaceae or Nonfermenters (of glucose) to differentiate them from the Enterobacteriaceae. Several of the bacteria in this group include bacteria such as Pseudomonas aeruginosa, Alcaligenes faecalis and Acinetobacter baumanii. These bacteria are mostly found in the environment, yet some of these organisms can cause wound infections and serious life-threatening infections in immunocompromised patients.
The Enterobacteriaceae have four common characteristics:
- They are all Gram-negative bacilli
- They all ferment glucose
- They do not produce oxidase
- They all reduce nitrate to nitrite
The Non-Enterobacteriaceae also have three common characteristics:
- They are all Gram-negative
- They do not ferment glucose
- Many (but not all) are oxidase positive
PRE-ASSESSMENT
PROCEDURE
For this exercise: Work in groups of 4. Each person in the group will work with one of the color dot cultures.
NITRATE REDUCTION TEST
Nitrate broth is used to determine if an organism can reduce nitrate. Some bacteria can reduce nitrate (NO3) to nitrite (NO2) by producing the enzyme nitrate reductase. Other bacteria can reduce nitrate to nitrogen gas by also producing the enzyme nitrite reductase which reduces nitrite to nitrogen gas. Other organisms do not have the ability to reduce nitrate at all.
Nitrate -> Nitrite -> Nitrogen Gas
1. Obtain a nitrate broth. Make a tape label writing your name, your assigned culture and the name of the media.
2. Using a sterile inoculating loop, obtain a small amount of bacteria and inoculate the nitrate broth.
3. Incubate the nitrate broth in the class test tube rack.
AFTER INCUBATION –Add reagents to the incubated nitrate broth. Add 10 drops of 0.8% sulfanilic acid and then add 10 drops of 0.6% N, N-Dimethyl-alpha-naphthylamine. IMMEDIATELY look for a dark red color which indicates the organism reduced nitrate to nitrite. This is a positive test result, and no further testing is required. Record your reaction and results on the worksheet.

IF no red color occurs: Dip a wooden applicator stick in the zinc powder (just enough to get the stick “dirty”) and drop it into the nitrate broth. If the organism reduced the nitrate to nitrogen gas, then no color will develop after the addition of zinc. This is considered a positive result since the organism reduced the nitrate to nitrogen gas. If the organism did not reduce nitrate, then the broth will turn red AFTER the addition of zinc because zinc reduces the nitrate to nitrite, NOT the bacteria. A red color formation after the addition of zinc is a negative test result. Record your reaction and results on the worksheet.
After adding sulfanilic acid & alpha-naphthylamine
Red color = Positive (Nitrate was reduced to Nitrite)
If no color, then add zinc
No color = Positive (Nitrate was reduced to Nitrogen gas)
Red color = Negative (Nitrate was not reduced by the bacteria)

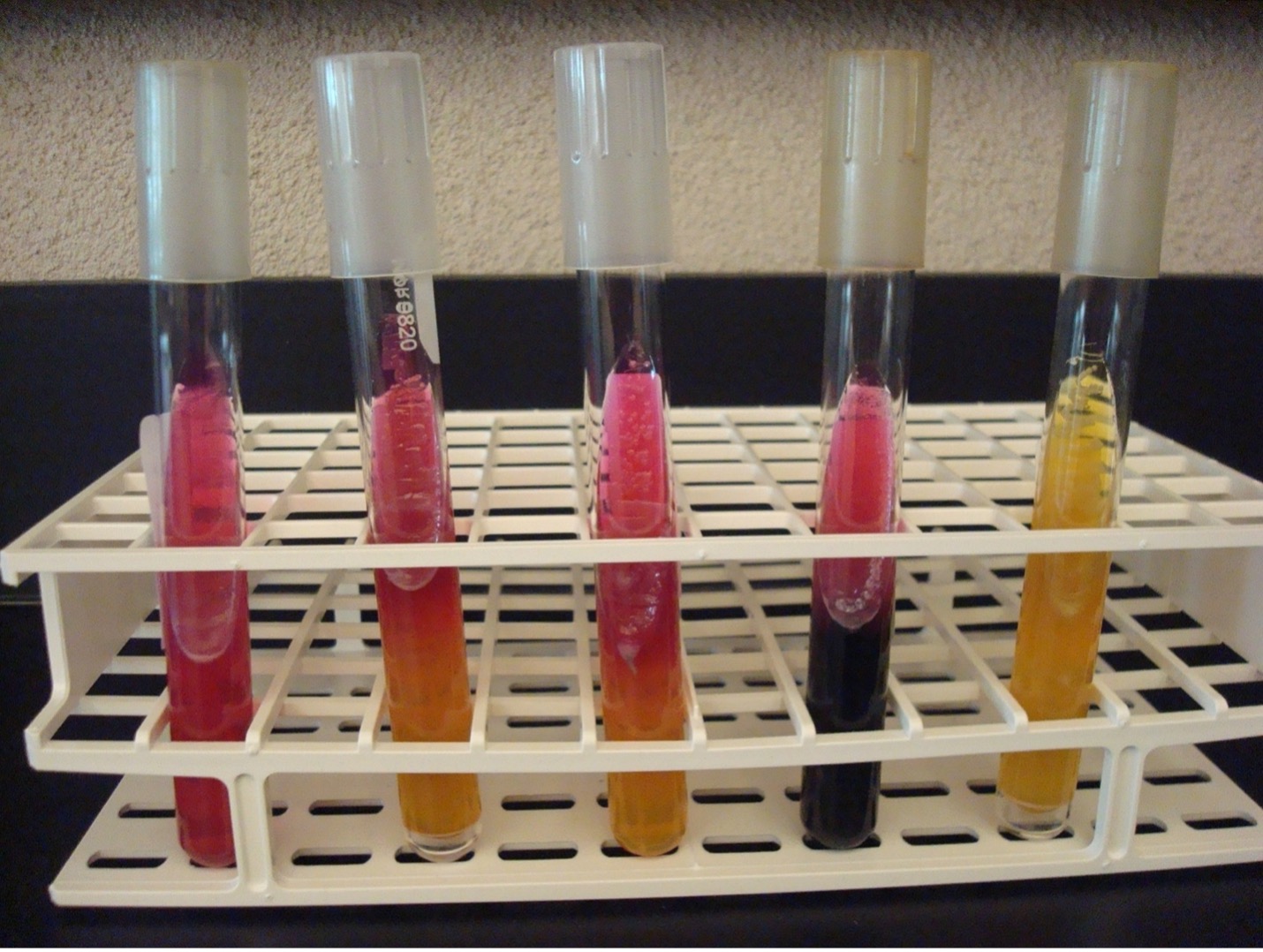
KLIGLER IRON AGAR (KIA)
KIA media contains two carbohydrates, glucose and lactose. It also has a phenol red pH indicator, ferrous sulfate, and peptones in the agar.
KIA media is used to determine if the organisms can ferment glucose with or without gas production. If the organism can ferment glucose, then KIA will also demonstrate if it can ferment lactose. The media can also determine if the organism can produce hydrogen sulfide.
If bacteria cannot ferment glucose, they are called “nonfermenters” and will not be able to ferment any other carbohydrates.
If bacteria can ferment glucose, they are called “fermenters” and can be differentiated by determining if it can also ferment lactose or produce hydrogen sulfide.
1. Obtain a KIA tube and a straight inoculating wire. Label the tube with your name, your assigned culture and the name of the test.
2. Use the sterile straight inoculating wire to take an inoculum of the bacteria.
3. Begin by stabbing in the middle of the KIA slant to the bottom of the tube. As the needle is withdrawn from the agar, streak the agar slant.
4. Incubate the KIA tube in the class test tube rack.
AFTER INCUBATION –Observe the reactions in the KIA tube and record the reactions/results. Record the reactions and results on the worksheet.
Butt of KIA tube:
Yellow butt = Positive glucose fermentation (A black precipitate may mask the yellow color)
Red butt = Negative glucose fermentation
Gas production from glucose fermentation:
Bubbles or splitting of the agar = Positive for gas (If bacteria cannot ferment glucose, they will not produce gas)
Slant of the KIA tube:
Yellow slant = Positive lactose fermentation
Red slant = Negative lactose fermentation
Hydrogen sulfide production:
Black precipitate in the media = Positive Hydrogen Sulfide Production
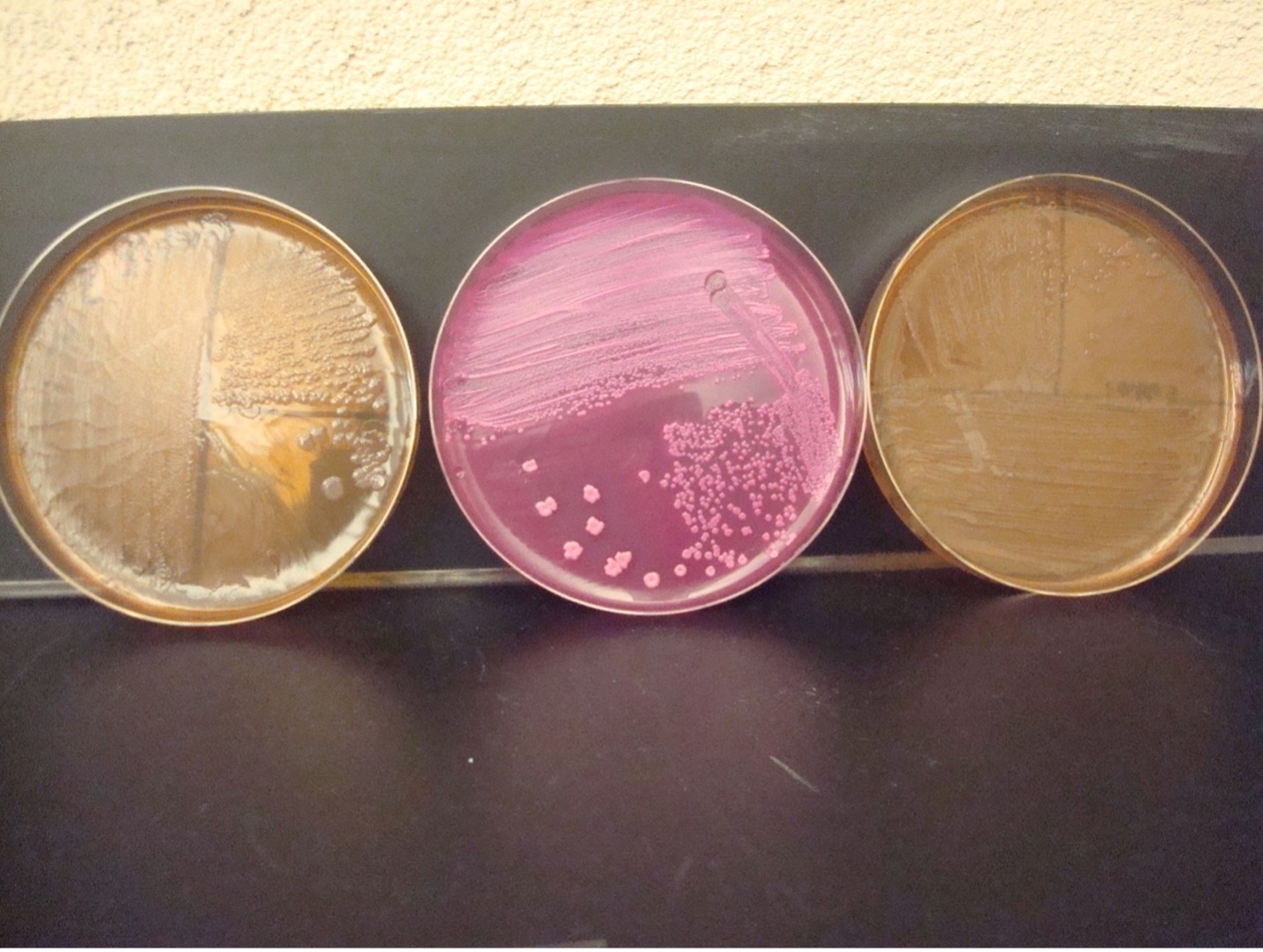
MACCONKEY AGAR (MAC)
MAC agar is a selective and differential media. It is used to grow Gram-negative rods and differentiate their ability to ferment lactose. MAC has bile salts and crystal violet dye which inhibit Gram-positive bacteria. The media also contains the carbohydrate lactose and a pH indicator. The Gram-negative rods that ferment lactose will produce acidic end products thereby changing the color of the pH indicator from clear to hot pink.
1. Obtain a MacConkey agar (MAC) and label the agar side with your name, your assigned culture and the name of the media.
2. Using a sterile inoculating loop, take a small amount of the bacteria and streak for isolation on the MacConkey Agar plate.
3. Incubate the MAC in the class plate tray until the next laboratory session.
AFTER INCUBATION –Observe your MacConkey Agar plate. Record the appearance of the growth of the bacteria on this agar. Record the reactions and results on the worksheet.
Growth on media = Gram-negative bacilli
Determine the ability to ferment lactose:
Hot pink colonies = Positive for lactose fermentation
Clear colonies = Negative for lactose fermentation (some lactose negative colonies may have a slight pink color due to the transmittance of the media color through the colony)
OXIDASE TEST
This test determines if the bacteria can produce the enzyme cytochrome oxidase which transfers electrons in the electron transport chain in cellular respiration. The organism to be tested must be grown on non-inhibitory media such as KIA or TSA because the production of oxidase is easily inhibited by selective media.
1. Cut an oxidase strip in half with scissors. Place the oxidase strip face up (look at the edges of the strip, they should be pointing down) on a glass slide. Barely moisten the oxidase strip with a small drop of water. Do not over-moisten the strip!
2. Using a sterile swab, obtain a large amount of inoculum from the KIA or TSA slant. Rub the bacteria on the swab on the oxidase strip.
3. Watch for purple color to develop on the strip or swab within 20 seconds. Record the reaction/result on the worksheet.
Purple color change = Positive
No color change within 20 seconds = Negative

BIOCHEMICAL TESTS RESULTS OF GRAM NEGATIVE BACILLI
| TEST | Escherichia coli | Proteus mirabilis | Pseudomonas
aeruginosa |
Alcaligenes faecalis |
| Oxidase | Negative | Negative | Positive | Positive |
| Nitrate reduction | Positive
(NO3 ->NO2) |
Positive
(NO3->NO2) |
Positive
(NO3->N2) |
Negative
(NO3) |
| KIA Slant | Lactose Positive | Lactose Negative | Lactose Negative | Lactose Negative |
| KIA Butt | Glucose Positive | Glucose Positive | Glucose Negative | Glucose Negative |
| KIA Gas from fermentation | Positive | Not reliable | Negative | Negative |
| KIA H2S | Negative | Positive | Negative | Negative |
| Lactose fermentation on MacConkey agar | Positive | Negative | Negative | Negative |
POST TEST
DISCOVERIES IN MICROBIOLOGY
DR. EMIL von BEHRING
In 1890, German physiologist Dr. Emil von Behring published an article with Dr. Kitasato Shibasaburō reporting that they had developed “antitoxins” against both diphtheria and tetanus. They had injected diphtheria and tetanus toxins into guinea-pigs, goats and horses. The animals developed antitoxins (now known to contain antibodies) in their serum. These antitoxins could protect against and cure the diseases in non-immunized animals. In 1892 Dr. Behring began the first human trials of the diphtheria antitoxin, but they were unsuccessful. After the production and quantitation of the antitoxin was improved, in 1894 humans were successfully treated with the antitoxin.


