7.3 Narrowing It Down: Index Fossils and Absolute Dating
In the previous section we learned how we can put different rock layers and events such as unconformities, volcanic intrusions, and fault lines in the correct order from youngest to oldest in what is called Relative Dating. However, whenever it is possible, geologists prefer to narrow down the age of a certain rock layer as closely as possible. For example, while it is useful to know that a sandstone layer is younger than a shale layer below it and older than a conglomerate layer above it, a scientist would much prefer to know that the layer is 52.4 Ma in age –it’s useful to be precise!
In this section, we will look at two tools that geologists employ to find that sort of precision – index fossils and radiometric dating!
Index Fossils
Fossils are the hard remains, impressions, or traces of an organism that was once alive a long time ago. The example that we often think about are dinosaur bones – these bones have undergone remineralization, or a change where the minerals and organic matter once present in their structure have undergone recrystallization into hard materials. The same is true of other fossilized remains we encounter.

Some of the fossils we find in the geologic record are of organisms that only lived during a short period on Earth. Let’s consider a species of ammonite, which is a shelled spiral-shaped organism that went extinct with the dinosaurs. One species of the ammonite was called Tropites subbullatus, and it is found in many different parts of the world. However, this organism only lived during the specific time between 248 to 206 million years ago.
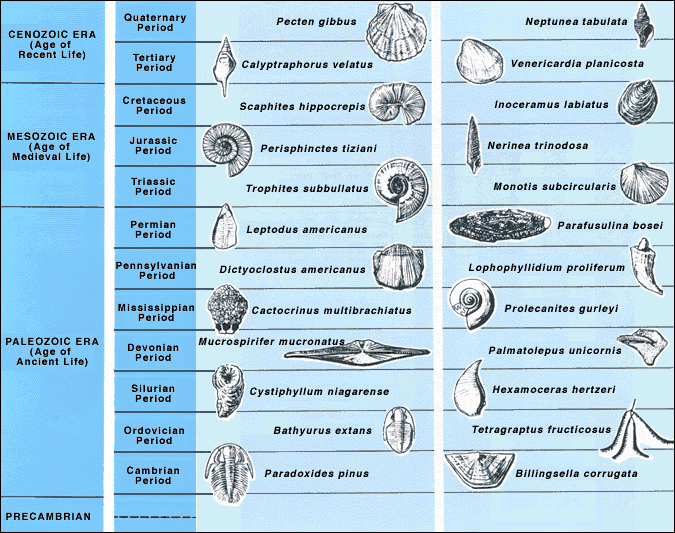
How is this useful? Well, if we were to encounter some limestone that had this fossil in it, we would instantly know that the limestone would have formed during the time this organism was alive – between 248 to 206 Ma. Knowing index fossils helps us determine the range in which rock layers formed.
The best index fossils are widespread, abundant, easily identifiable, and belong to an organism that lived during a very specific time in the geologic record. If the ammonite Tropites subbullatus was useful in determining that a rock layer was formed between 248 and 206 Ma, just imagine how much more useful multiple index fossils can be from around the same time period in narrowing down the rock layer’s age even further!
Try the exercise below to further understand the usefulness of multiple index fossils!
Absolute/Radiometric Dating
Technically, using index fossils is a method of Absolute Dating, “Absolute” dating essentially means assigning a specific chronological age to a rock layer. When we have multiple index fossils as evidenced by the previous exercise, we can narrow down an absolute age for a rock layer very well.
Another method that can determine the absolute age of a rock layer or layers is through radiometric dating. This method uses unstable, or radioactive isotopes, such as Uranium in the example of determining the age of the planet mentioned at the beginning of this chapter.

What is an isotope? It is a version of an element that contains a different number of neutrons in its nucleus. Let’s consider the basic example of hydrogen. Hydrogen has 1 proton in its nucleus and no neutrons – this is called Hydrogen-1. There are isotopes of Hydrogen with 1 proton and 1 neutron in its nucleus (Hydrogen-2, deuterium) and Hydrogen with 1 proton and 2 neutrons in its nucleus (Hydrogen-3, tritium).
Sometimes an isotope version of an element is unstable – these unstable isotopes are radioactive, and they naturally undergo decay to return to a more stable state. This decay occurs through the loss of a helium nucleus (2 protons and neutrons), known as an alpha particle, to produce a more stable “daughter” isotope or element.

The time it takes for the unstable radioactive isotope to decay into a stable daughter product depends on the element. For just half of Uranium-238 to decay into Lead-206, it takes 4.5 billion years.

Another useful pair in geology is Potassium-40, which decays into Argon-40. Half of it decays, in what is called the “half-life” in 1,250 million years. The Potassium/Argon system is particularly useful in dating volcanic and ash deposits in rock layers. Watch this useful video below to better understand this process!

