13.2 Soil Horizons
Katherine J. Megivern
Soil Horizons
The process of soil formation generally involves the downward movement of clay, water, and dissolved ions, and a typical result of that movement is the development of chemically and texturally different layers known as soil horizons. The typically developed soil horizons are listed below, followed by an idealized diagram of soil horizons that includes their (very) approximate relative thicknesses.
- O — the layer of organic matter
- A — the layer of partially decayed organic matter mixed with mineral material
- E— the eluviated (leached) layer from which some of the clay and iron have been removed to create a pale layer that may be sandier than the other layers (not shown in the first diagram)
- B — the layer of accumulation of clay, iron, and other elements from the overlying soil
- C — the layer of incomplete weathering of the parent material
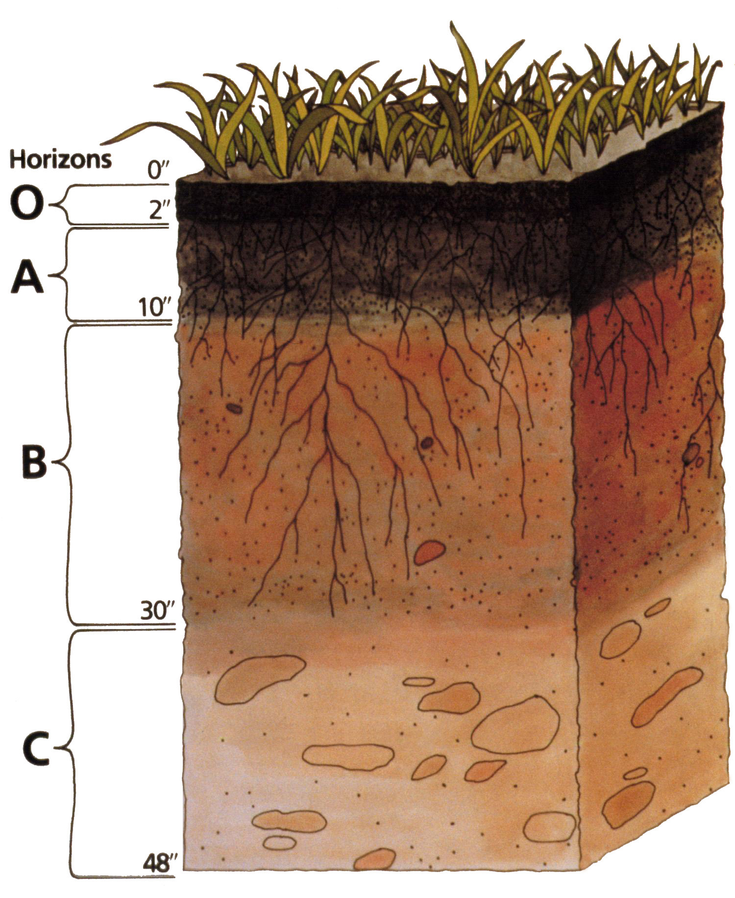
Figure 13.2.2. Soil horizons in a podsol (or podzol) soil type from a site in northeastern Scotland. The white layer under the humus is characteristic of this type of soil. Note that not all soils show layers as distinctly as this one. Key to layers: O: organic matter A: organic matter and mineral material E: leached layer B: accumulation of clay, iron etc. C: incomplete weathering of parent material
Another type of layer develops in hot, arid regions known as caliche (pronounced ca-lee-chee). It forms from the downward (or in some cases upward) movement of calcium ions, and the precipitation of calcite within the soil. When well developed, caliche cements the surrounding material together to form a layer that has the consistency of concrete. Needless to say, such layers can form a barrier to plant roots and water movement. Other times, caliche can form nodules as it precipitates on a nucleus of some type. Plant roots sometimes act as a nucleus and long lumpy nodules result. Pioneer children, finding these “loess kindchen” (loess children, translated) in deposits of loess (windblown silt) are said to have used them as dolls.

Soil Nutrients
Soil is an important reservoir for organic components necessary for plants, animals, and microorganisms to live. The organic component of soil, called humus, is a rich source of bioavailable nitrogen. Nitrogen is the most common element in the atmosphere, but it exists in a form most life forms are unable to use. Special bacteria found only in soil provide most nitrogen compounds that are usable by life forms (“bioavailable”) through a process termed nitrogen fixation. Follow the routes of nitrogen in the diagram below and refer to the diagram as you read the description below the diagram.
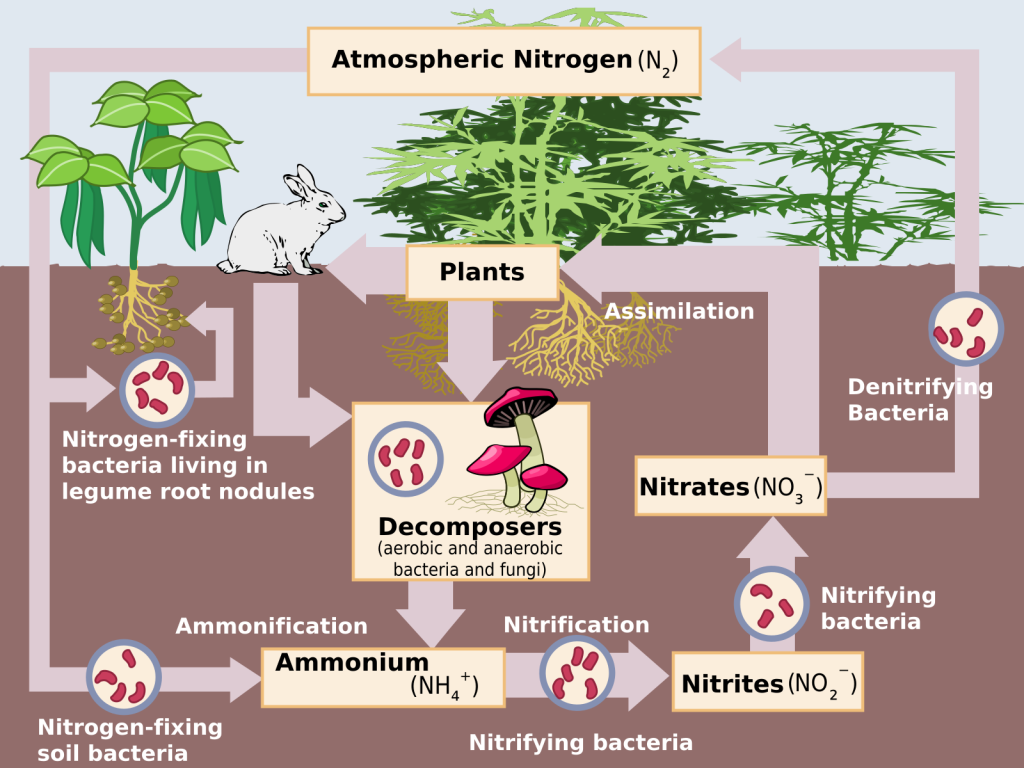
The nitrogen-fixing bacteria absorb nitrogen from the atmosphere and convert it into nitrogen compounds. These compounds are absorbed by plants and used to make DNA, amino acids, and enzymes. Animals obtain bioavailable nitrogen by eating plants, and this is the source of most of the nitrogen used by life. That nitrogen is an essential component of proteins and DNA.
Soils range from poor to rich, depending on the amount of humus they contain. Soil productivity is determined by water and nutrient content, with nitrogen and phosphorus being two key nutrients in soils.
Video 13.2.1. Nitrogen & Phosphorus cycles: Always Recycle! Part 2. (9:21) This is a fun, fast-paced, super-informative video on two critical biogeochemical cycles. https://www.youtube.com/watch?v=leHy-Y_8nRs&t=16s&ab_channel=CrashCourse
Freshly created volcanic soils, called andisols, and clay-rich soils that hold nutrients and water are examples of productive soils. The CEC, or cation exchange capacity, of soils is a common measure of soil productivity in terms of their potential to deliver important positively-charged ions such as calcium, magnesium, and potassium to locations near the plant roots.
Video 13.2.2. Cation Exchange. (5:49) The importance of clay in getting nutrients to plants and how this relates to fertilizers.
The beginning of the chapter mentioned that it should take a paragraph to define soil…perhaps a single paragraph won’t be enough…
A charged atom or molecule.


