6.1 Distribution of Earth’s Water
Merry Wilson
Water, Water, Everywhere!
Water is composed of two atoms of hydrogen and one atom of oxygen bonded together. Despite its simplicity, water has remarkable properties. Water expands when it freezes. It has high surface tension because of the molecules’ polar nature that tend to stick together. Without water, life might not exist on Earth, and it certainly would not have the tremendous complexity and diversity that we see (1). Video 6.1.1. explains the polarity and the properties that arise.
Video 6.1.1. Water is both essential and unique. Many of its particular qualities stem from its polarity, thanks to two hydrogen atoms and one oxygen. (3:51)
Where does all the Earth’s water come from? Primordial Earth was an incandescent globe of magma, but all magmas contained water in them. Water was released as water vapor from magma and it accumulated in Earth’s atmosphere. When the temperature decreased, the water vapor condensed and precipitated as rain, accumulating on the surface as liquid water. Volcanic activity kept, and still keeps, introducing water into the atmosphere, thus increasing the surface- and groundwater volume of the Earth (2).

Earth’s oceans contain 97% of the planet’s water, and just 3% is freshwater with relatively low concentrations of salts. Most freshwater is trapped as ice in the vast glaciers and ice sheets of Greenland and Antarctica. A storage location for water such as an ocean, glacier, pond, or even the atmosphere is called a reservoir. A water molecule may pass through a reservoir quickly or may remain for much longer. As you can see from the graph below, Rivers account for 0.49% of surface/ other freshwater or 0.0001% of the overall water on the planet (1).
Hydrologic Cycle
Because of the unique properties of water, water molecules can cycle through almost any-where on Earth. The water molecule found in a glass of water today could have erupted from a volcano early in Earth’s history. In the intervening billions of years, the molecule probably spent time in a glacier or far below the ground. The molecule surely was high in the atmosphere and maybe deep in the belly of a dinosaur.
Water is the only substance on Earth that is present in all three states of matter—as a solid, liquid, or gas. Along with that, Earth is the only planet where water is present in all three states. Because of the ranges in temperature in specific locations around the planet, all three phases may be present in a specific location or region. The three phases are solid (ice or snow), liquid (water), and gas (water vapor) (Dastrup, 2020).
Video 6.1.2. Block diagram animations of the water cycle (1:22)
The water cycle has no starting point. But we’ll begin in the oceans, since that is where most of Earth’s water exists. The sun, which drives the water cycle, heats water in the oceans. Some of it evaporates as vapor into the air. Ice and snow can sublimate directly into water vapor. Rising air currents take the vapor up into the atmosphere, along with water from evapotranspiration, which is water transpired from plants and evaporated from the soil. The vapor rises into the air where cooler temperatures cause it to condense into clouds (USGS).
Air currents move clouds around the atmosphere, cloud particles collide, grow, and fall out of the sky as precipitation. Some precipitation falls as snow and can accumulate as ice caps and glaciers, which can store frozen water for thousands of years. Snowpacks in warmer climates often thaw and melt when spring arrives, and the melted water flows overland as snowmelt (USGS).
Most precipitation falls back into the oceans or onto land, where, due to gravity, the precipitation flows over the ground as surface runoff. A portion of runoff enters rivers in valleys in the landscape, with streamflow moving water towards the oceans. Runoff, and groundwater seepage, accumulate and are stored as freshwater in lakes. Not all runoff flows into rivers, though. Much of it soaks into the ground as infiltration. Some water infiltrates deep into the ground and replenishes aquifers (saturated subsurface rock), which store huge amounts of freshwater for long periods of time (USGS).
Some infiltration stays close to the land surface and can seep back into surface-water bodies (and the ocean) as groundwater discharge, and some groundwater finds openings in the land surface and emerges as freshwater springs. Over time, though, all of this water keeps moving, some to reenter the ocean, where the cycle continues (USGS).
And, of course, it’s complicated….
Video 6.1.3. This animation uses Earth science data from a variety of sensors on NASA Earth observing satellites and cartoons to describe Earth’s water cycle and the continuous movement of water on, above and below the surface of the Earth (5:52).
People also depend on water as a natural resource. Not confined to only take fresh water directly from streams or ponds, humans create canals, aqueducts, dams, and wells to collect water and direct it to where they want it, which further complicates the already dynamic system.
Backyard Geology: Arizona’s water cycle
Arizona is the sixth largest state in the US, and it encompasses diverse climates and topography.
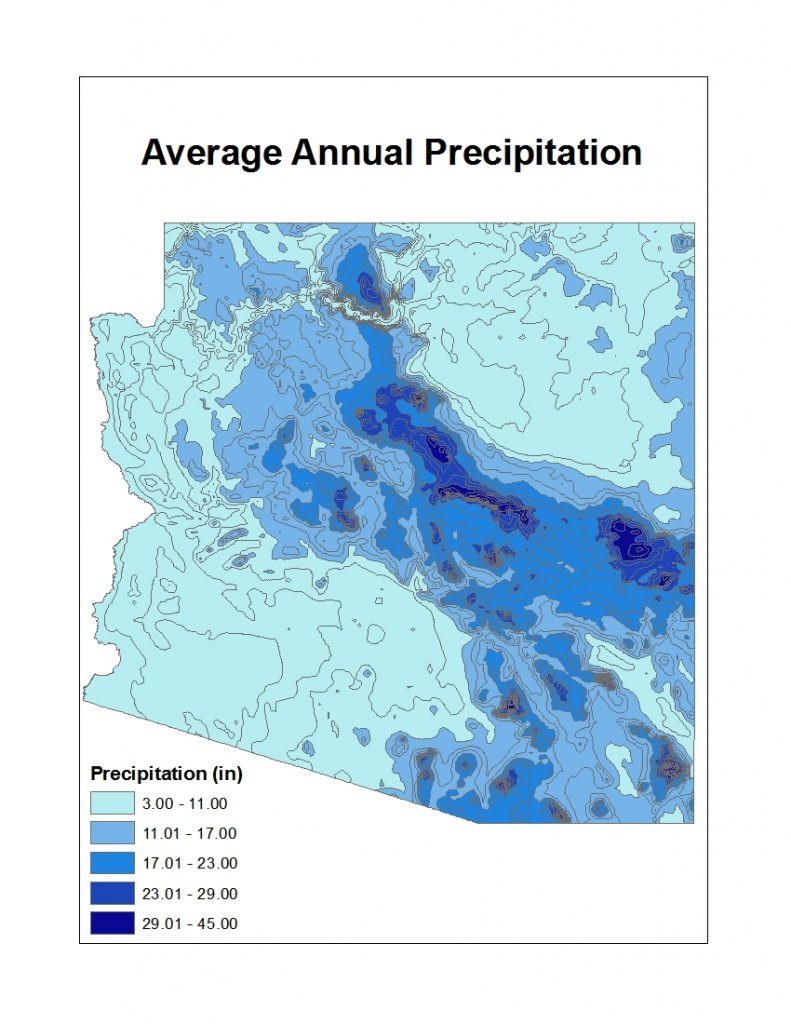
Precipitation: Much of the state is characterized as arid to semi-arid with total annual precipitation ranging from less than 3 inches in the southwest to around 40 inches in the White Mountains in east central Arizona. Precipitation is highly variable from year to year, with statewide average precipitation ranging from a low of 6.04 total inches in 1956 to a high of 22.77 inches in 1905.
Evaporation: It is estimated that more than 95% of rainfall that falls in the non-mountainous regions of Arizona evaporates quickly. The largest natural lake in Arizona is Mormon Lake, near Flagstaff, AZ, and has been known to be completely dry.
Runoff (Streams): This is extremely variable, and generally related to weather patterns and rainfall through storms. Most streams in Arizona either infiltrate into the groundwater, evaporate, or flow to the Colorado River. All major streams have at least one dam on them, and very little water flows all the way to the ocean.
layer of gases that surrounds a planet
a state of matter that can flow and will fill low spaces of a container
The largest reservoir of water on earth, containing over 96% or all Earth's water. This water is saline (saltwater).
The conversion between solid and gaseous phases of matter, with no intermediate liquid stage.
The total of water evaporated from the land surface plus transpiration from plants
proccess of giving off water vapor through openings in leaves
process of evaporation that changes liquid and frozen water into water-vapor gas, which then floats up into the skies to become clouds.
Condensation is the process of water vapor turning back into liquid water, with the best example being clouds.
When the cloud droplets combine to form heavier cloud drops which can no longer "float" in the surrounding air, it can start to rain, snow, and hail... all forms of precipitation, the superhighway moving water from the sky to the Earth's surface.
precipitation that flows out over the landscape
water soaking into the ground
water that emerges onto the land surface into rivers and lakes
water filling pore spaces beneath the ground surface
a place where water moving underground finds an opening to the land surface and emerges, sometimes as just a trickle, maybe only after a rain, and sometimes in a continuous flow

